Plasticity induced by nanoindentation in CrCoNi medium-entropy alloy studied by accurate electron channeling contrast imaging revealing dislocation-low angle grain boundary interactions
Materials Science and Engineering A, 2021, 817, 141364 DOIElementary plasticity mechanisms and dislocation-interface interactions
No crystal is perfect. The tiny imperfections hidden inside metals and ceramics — called defects — are actually what control how strong, conductive, or brittle a material is. We study these defects to understand why materials behave the way they do.
What is a crystal defect?
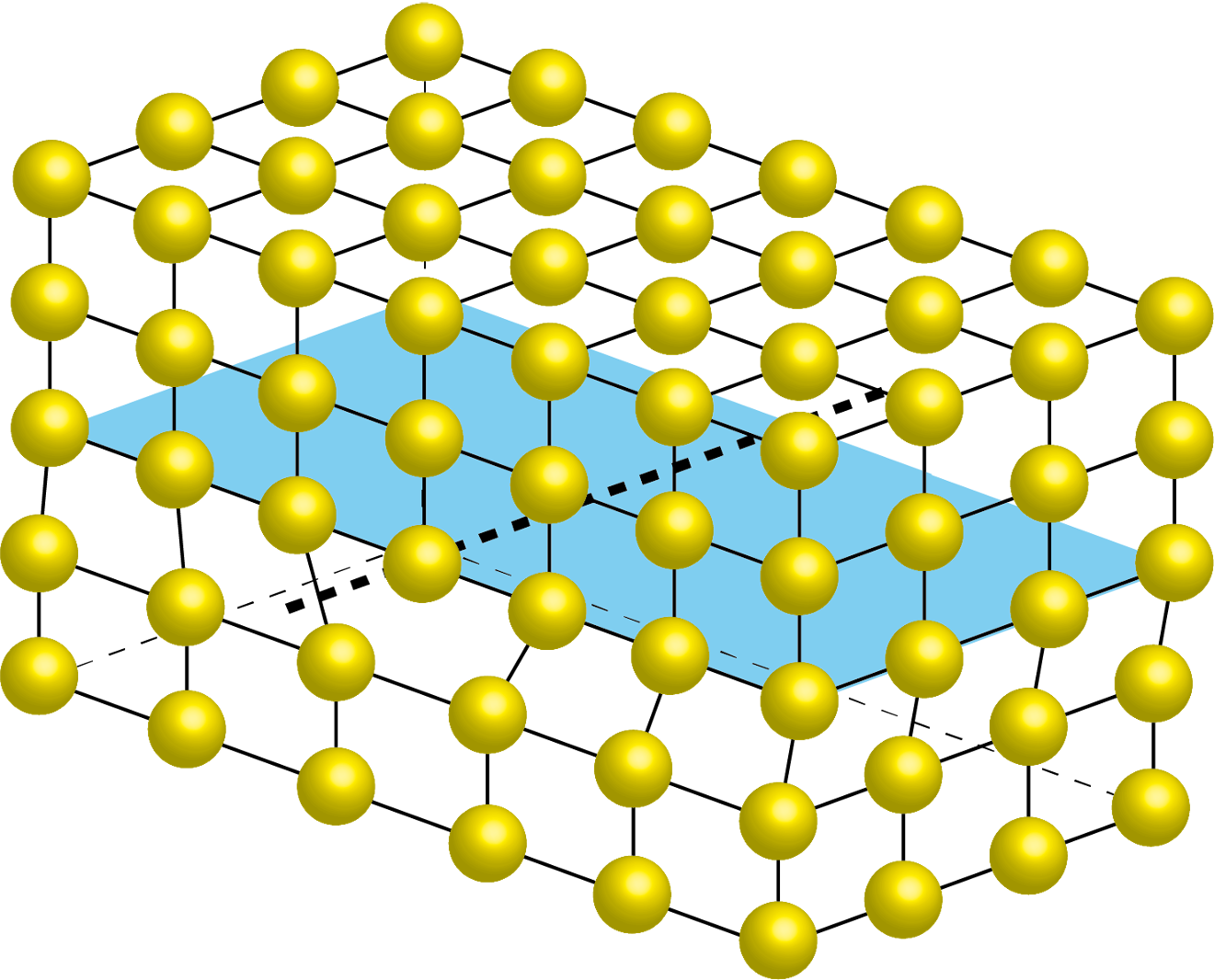
Imagine building a perfectly regular wall of bricks — same size, same spacing, every row aligned. Now imagine one brick is missing, one is slightly tilted, or two rows have slipped past each other. These imperfections are what physicists call crystal defects: disruptions in the otherwise perfectly ordered arrangement of atoms inside a solid material.
At first glance, defects might sound like a problem to be avoided. In reality, they are what make materials useful. The strength of steel, the electrical conductivity of semiconductors, the ability of a metal to absorb radiation — all of these properties are governed not by the perfect crystal, but by its imperfections.
"Crystals are like people; it is the defects in them which tend to make them interesting!"
— Colin J. Humphreys, PhysicistA family of imperfections
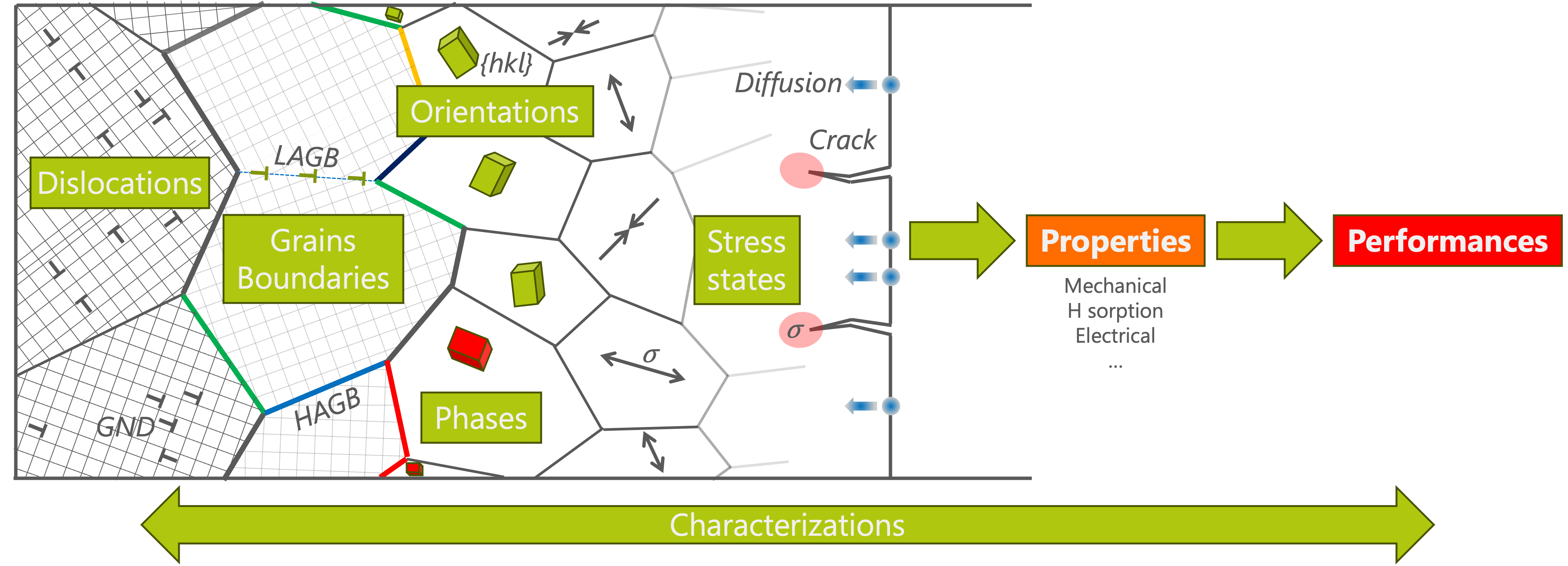
Crystal defects come in several flavours, depending on how many dimensions they disrupt:
- Point defects — a single atom is missing (vacancy), replaced by a foreign atom (substitution), or squeezed in where it shouldn't be (interstitial). These are the simplest defects, yet they drive phenomena like hydrogen storage and irradiation damage.
- Line defects (dislocations) — a whole line of atoms is misaligned. When a metal bends without breaking, it is because dislocations are moving through it. They are the microscopic mechanism behind plastic deformation.
- Planar defects (grain boundaries) — a flat interface between two regions of crystal with slightly different orientations. Grain boundaries act as barriers to dislocation motion, which is why fine-grained metals are generally stronger.
- Extended defects — larger 3D disruptions such as precipitates or voids, often introduced by irradiation or thermal treatments.

Dislocations: the engines of deformation
Of all defect types, dislocations are the ones my group studies most closely. A dislocation is essentially an extra half-plane of atoms inserted into the crystal lattice — like a crease in a rug. When you push the rug, the crease moves across the room far more easily than if you tried to slide the entire rug at once. In the same way, dislocations allow metals to deform plastically at stresses far lower than a perfect crystal would require.
Understanding how dislocations nucleate, multiply, interact, and eventually pile up or become tangled is central to predicting when a metal will yield, harden, or fracture.
Why does it matter?
By understanding the link between defects and properties, we can engineer materials with purpose. The questions we ask are practical ones: How can we make a metal harder without making it brittle? How do defects change when a material is irradiated in a nuclear reactor? Why does a semiconductor lose efficiency as its dislocation density increases?
My research tackles these questions across a range of materials — metals, ceramics, intermetallics, semiconductors — and a range of properties: mechanical strength, hydrogen storage capacity, irradiation resistance, and electrical behaviour. The common thread is always the defect, observed at the atomic scale, understood in its physical context, and ultimately harnessed to improve real-world performance.
Selected publications
A selection of publications from this research axis: